The purpose is to ensure that personnel are familiar with the proper procedures for protecting vacuum lines and staff from potentially hazardous biological materials. Some labs may be functioning under the misconception that if they use one flask that contains disinfectant, the lines are adequately protected. This is not the case.
The University of Toronto Biosafety Policies and Procedures Manual is available via the U of T Biosafety web site.
This document requires that: “Vacuum lines used for work involving the (biological) agent must be protected from contamination by HEPA filters or equivalent equipment.”
A recommended procedure is described below. It requires the use of a cartridge-type filter, which is easily installed and prevents the passage of aerosols through the vacuum line.
If personnel in your lab use hazardous biological materials (e.g. human cell cultures, viral vectors, pathogenic microorganisms or potentially hazardous proteins) in conjunction with vacuum systems would you please ensure the vacuum lines and staffs are protected in accordance with the following procedure.
If you have questions or need additional information, please contact the U of T Biosafety Officer, at (416) 978-3981. Thank you for your cooperation.
Procedures For Protecting Vacuum Lines
Vacuum line traps and filters prevent the suction of potentially hazardous biological materials into vacuum lines. The use of traps also helps keep vacuum lines from clogging. The following diagram depicts the proper set-up for vacuum lines that are used with potentially hazardous biological materials.
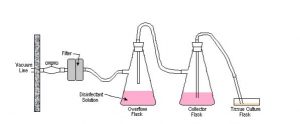
Note:
1. The collection flask should be routinely monitored, and emptied or replaced before it is filled. When using the vacuum line in a biological safety cabinet (tissue culture hood), place the collection flask inside the cabinet (hood). The second (overflow) flask can be located outside the cabinet. However, never use a glass flask at floor level unless a sturdy, non-glass container adequately shields it.
2. Start with full strength chemical disinfectant in the collection flask. Allow the aspirated fluids to dilute the disinfectant, until it is at the recommended dilution. Following a contact period to allow disinfection, discard the solution. For example: start with 100 mL of bleach; aspirate waste fluids; ensure that a final of 1% sodium hypochlorite is achieved in your waste solution; discard after a sufficient contact period.
3. Replace the vacuum filter when it is clogged or if liquid makes contact with it.
4. Check that all connections and seals are tight to assure the vacuum is adequate